Subscribe to Pittwire Today
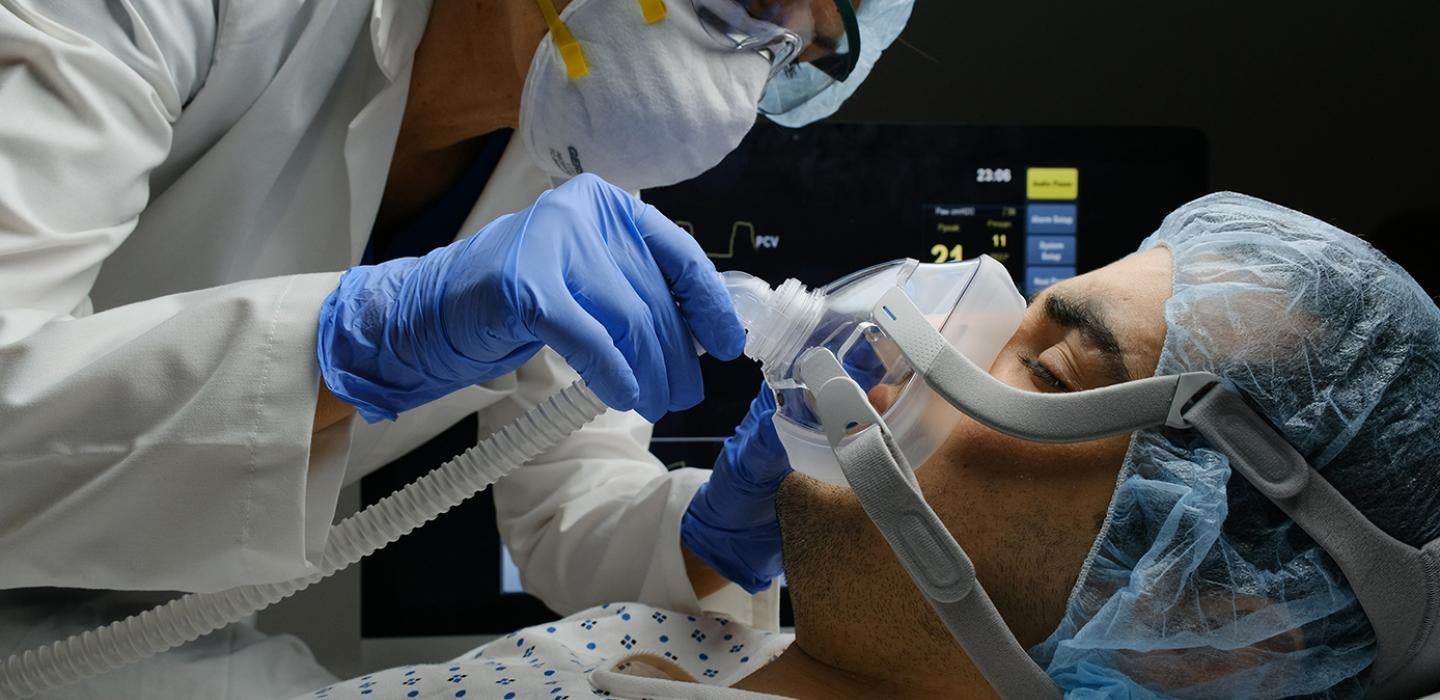
Get the most interesting and important stories from the University of Pittsburgh.In a demonstration of global collaboration, an international team led by clinician-scientists at the University of Pittsburgh School of Medicine and UPMC have pooled data from 121 hospitals in eight countries to find that inexpensive, widely available steroids improve the odds that very sick COVID-19 patients will survive the illness.
The findings were made through the “Randomized Embedded Multifactorial Adaptive Platform-Community Acquired Pneumonia” (REMAP-CAP) trial and are reported today in JAMA as part of a four-article package. The World Health Organization is updating its COVID-19 treatment guidance as a result.
“It is relatively rare in medicine that you find drugs where the evidence of their effectiveness in saving lives is so consistent,” said lead author Derek Angus, professor and chair of Pitt’s Department of Critical Care Medicine and chief health care innovation officer at UPMC. “This is, in many respects, the single clearest answer we’ve had so far on how to manage terribly ill COVID-19 patients. People on ventilators or oxygen and under intensive care should definitely be given corticosteroids.”REMAP-CAP includes the UPMC-REMAP-COVID19 trial, the only U.S.-based trial to test corticosteroids—a class of drug that lowers inflammation and modulates immune system activity—for treating critically ill COVID-19 patients.
Between March and June, the REMAP-CAP corticosteroid trial randomized 403 adult COVID-19 patients admitted to an intensive care unit to receive the steroid hydrocortisone or no steroids at all. The trial found a 93% probability that giving patients a seven-day intravenous course of hydrocortisone would result in better outcomes than not giving the steroid. The results were consistent across age, race and sex.
“This gives physicians like me, who treat the sickest of the sick, hope,” said co-author Bryan McVerry, associate professor of pulmonary, allergy and critical care medicine. “We are beginning to get a handle on the deadly side of this disease.”
REMAP-CAP and the other corticosteroid trials did not test the drugs in non-hospitalized patients with COVID-19 who did not need respiratory support. Steroids currently are not recommended for these patients because they can dampen the immune system and have serious side effects. In addition, the REMAP-CAP corticosteroid trial was mostly conducted in resource-rich countries across Europe, North America and Australasia, so the findings may not translate to low- and middle-income countries.
In the news
Read more about the research in:
Because it is designed to simultaneously test multiple combinations of potential therapies—as opposed to the traditional, slow clinical trial process that tests one therapy at a time—REMAP-CAP is particularly well-suited for rapidly identifying effective treatments during the COVID-19 pandemic. It is currently testing thousands of different treatment regimens, including various doses and combinations of vitamin C, convalescent plasma, blood thinners, antivirals and immune modulators.
“REMAP-CAP and our findings on corticosteroids are possible because of a global community of clinicians and scientists coordinating and sharing data across different languages and countries,” said co-author Christopher Seymour, director of the Translational and Clinical Science Program at the Clinical Research, Investigation and Systems Modeling of Acute Illness (CRISMA) Center in Pitt’s School of Medicine. “This is how we get definitive answers as fast as possible on how to best treat patients. Outcomes in Amsterdam are helping patients at UPMC Altoona.”
Timothy Girard, Christopher Horvat, David Huang, Kelsey Linstrum and Stephanie Montgomery, all of Pitt’s CRISMA Center, also contributed to this research.
Building on Pitt progress
Today’s discovery follows in Pitt’s storied history of innovation in steroid use. Alumnus Philip Showalter Hench (MED 1920) was awarded the 1950 Nobel Prize in Medicine for his discovery of the first steroid treatment, which was used for rheumatoid arthritis.
Additional efforts at Pitt to fight COVID-19 include work led by Paul Duprex in the Center for Vaccine Research (CVR) to harness a measles vaccine engineered to express SARS-CoV-2 proteins on its surface to generate immunity to the virus that causes COVID-19.
Also through CVR, William Klimstra, associate professor of immunology, is investigating an antibody therapy called SAB-185 that could be used to both treat COVID-19 and prevent it in frontline workers and in military personnel. In August, SAB-185 was injected into healthy volunteers for a phase 1 safety study.
As part of the national effort Operation Warp Speed, Pitt and UPMC have been chosen as a site for Moderna and AstraZeneca vaccine trials. The phase 3 trials, led by Judy Martin, codirector of the Pittsburgh Vaccine Clinical Trials Unit and professor of pediatrics, and Sharon Riddler, associate professor of medicine, seek to determine if the vaccines can prevent COVID-19 and for how long.
Additionally, Pitt’s Clinical and Translational Science Institute has awarded nearly $1 million to 17 studies working on different aspects of the COVID-19 pandemic, from modeling the virus to developing treatments and figuring out the role of the lung’s microbiome in the infection.
In all, there are more than 400 ongoing studies related COVID-19 at Pitt.