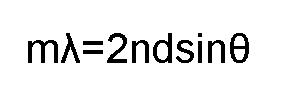Please
click on the topic you would like more information on:



Diffraction:
You
may be familiar with the phenomena of diffraction already and
not even be aware of it. You see forms of diffraction in many everyday
objects (see pictures below). We, in the Asher Research Group,
exploit this natural phenomena for developing novel materials for sensing and other
applications.
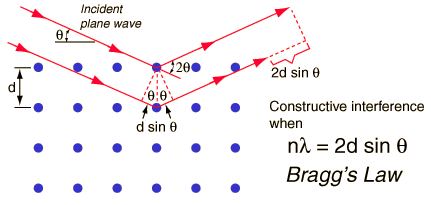
Bragg diffraction
was first proposed by William Lawrence Bragg in 1912 as a means
of analyzing the structure of crystals. Bragg and his father (William
Henry Bragg) collimated x-rays to diffract off of different crystal
planes. The x-rays were then collected in an ionization chamber
and the level of ionization was measured as a function of the
incident angle of the x-rays. Using this method, the Braggs were
able to determine the crystalline spacing for a number of substances.
The Bragg Condition
is given by:
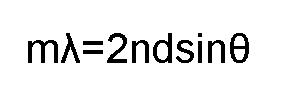
Waves that satisfy
this condition interfere constructively and have the same apparent
strength as reflection.
Photonic crystals
are periodic dielectric or metallo-dielectric (nano)structures
that are designed to affect the propagation of electromagnetic
waves. Since the basic physical phenomenon is based on diffraction,
the periodicity of the photonic crystal structure has to be in
the same length scale as the wavelength of the electromagnetic
waves (~300 nm for photonic crystals operating in the visible
part of the spectrum). Photonic crystals are attractive optical
materials for controlling and manipulating the flow of light.
They are of great interest both for fundamental and applied research.
back
to top



Crystalline
Colloidal Array Fabrication:
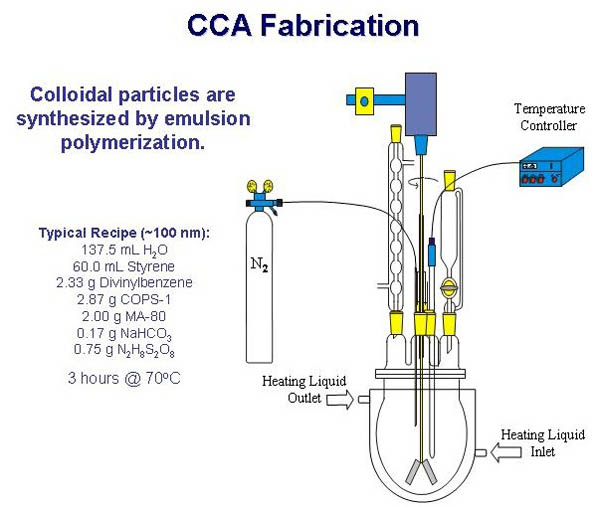
CCAs
are typically synthesized through a free-radical heterogeneous
nucleation emulsion polymerization. This synthesis can be used
to prepare particles that are 100 - 400 NM in diameter. The reactants
for the polymerization include an emulsifier, slightly water-soluble
monomers, a less water-soluble crosslinker, a water-soluble initiator,
and a buffer in an aqueous polymerization medium. The surface
charge and size of the resulting particles can be altered by varying
the relative amounts of these reactants.

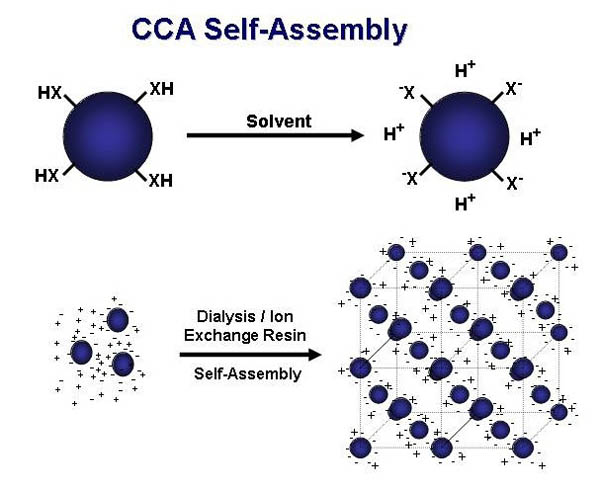
Aqueous suspensions
of monodisperse, highly-charged polystyrene particles self-assemble
into highly-ordered three-dimensional arrays, known as crystalline
colloidal arrays (CCAs). This self-assembling occurs in low ionic
strength media due to electrostatic repulsion, originating from
the ionization of sulfonate and carboxylate groups on the surface
of each sphere. The system minimizes its free energy by assembling
into either a face-centered cubic (FCC) or body-centered cubic
(BCC) lattice.
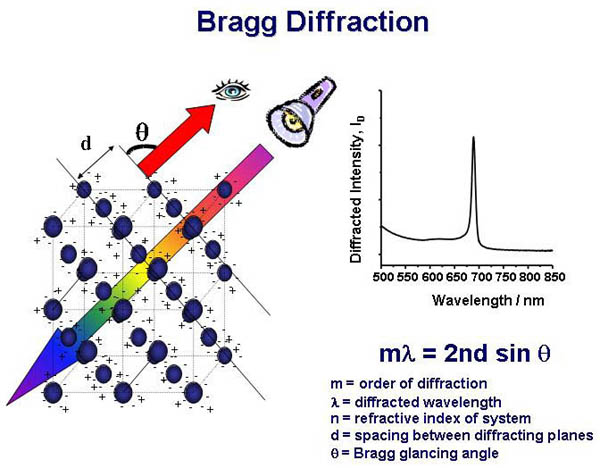
The periodic modulation
in the CCA refractive index and the lattice spacing of the array
are such that visible light is diffracted according to Bragg's
Law. The lattice spacing can be tuned, either by changing the
particle size or the particle concentration, so that CCAs can
efficiently diffract light in the near-UV, visible and near-IR
spectral regions. .
back
to top



Polymerized
Crystalline Colloidal Array Fabrication:
Since the ordering
of the CCA depends on the electrostatic repulsion between particles,
the lattice will disorder in the presence of ionic impurities.
The CCA lattice can be stabilized by polymerizing it within a
hydrogel to form a polymerized crystalline colloidal array (PCCA).
To form the PCCA, nonionic polymerizable monomers, cross-linkers
and photoinitiators are dissolved into the CCA. The mixture is
then polymerized within a quartz cell.

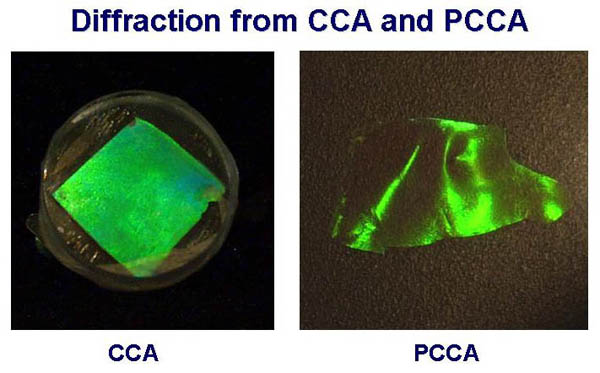
The resulting
PCCA is considerably more robust than the CCA. Also, since the
CCA lattice is embedded within the hydrogel, the observed diffraction
closely follows the hydrogel volume. As the hydrogel undergoes
a volume change, the spacing between the CCA particles will change,
resulting in a change in observed diffraction. To apply this motif
to chemical sensing, molecular recognition groups are incorporated
into the hydrogel backbone either during or after polymerization.
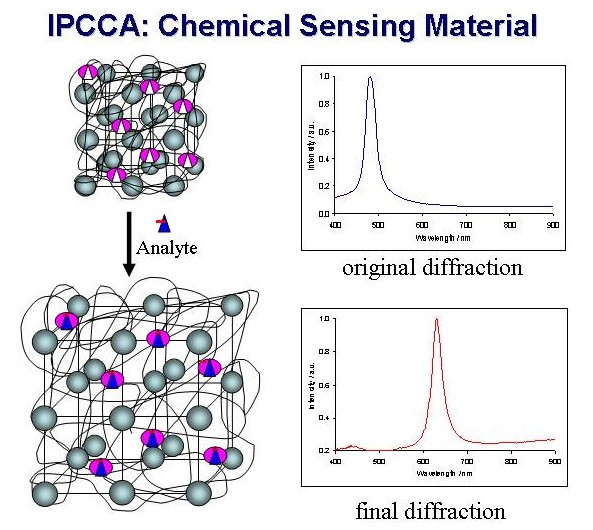
The resulting intelligent polymerized crystalline colloidal array
(IPCCA) will optically report on the concentration of the analyte
of interest. As the analyte of interest is bound, there will be
a change in the free energy of the system, resulting in a change
in the equilibrium volume of the hydrogel and a change in the
observed diffraction from the PCCA.
Back
to top



Some
Group Projects:
|
|
|
|
|
|
|
|
|
|
|
Development of 2-Dimensional
PCCA
|
|
Study of Interactions
between Nanoparticles within CCA
|
|
Method Development for
Measurement of Glucose Concentrations in Tear Fluid
|
|
Physiological pH Sensing
Material
|
|
Characterization of Mechanical
Properties of Nanocomposite PCCA Materials
|
|
Inverted-Structure Crystalline
Material
|
|
more to come soon
|
Glucose
Sensing Material
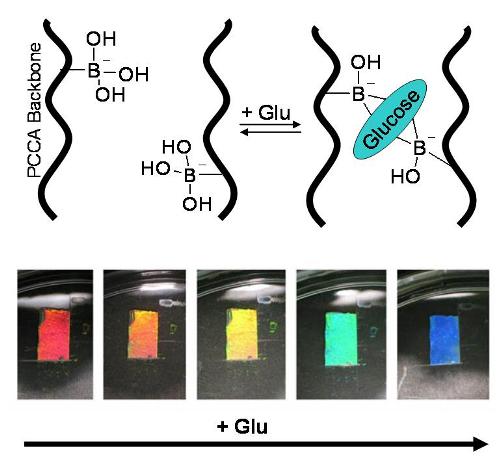
- We are developing a photonic
crystal glucose sensor consisting of a crystalline colloidal
array embedded within a polymer network with pendent phenylboronic
acid groups. The pendent boronic acid groups bind glucose
in a "sandwich-like" complex, forming additional crosslinks
in the hydrogel. As these additional crosslinks form, the
hydrogel shrinks and the diffraction blue-shifts in proportion
to the glucose in solution (see picture below).
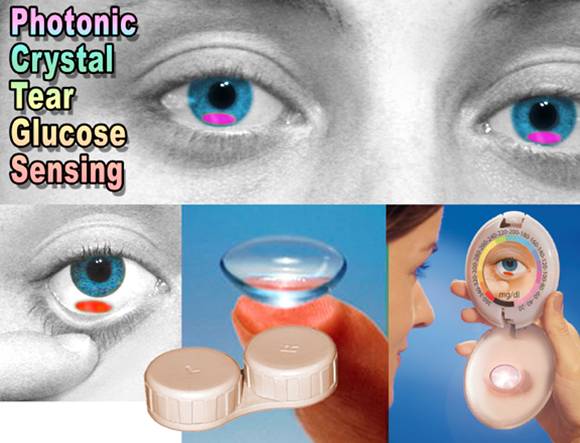
- We are optimizing our photonic
glucose sensing motif to sense glucose at the low glucose
concentrations of tear fluid. This sensing motif will be utilized
for the fabrication of noninvasive or minimally invasive in
vivo glucose sensing materials in the form of ocular inserts
or diagnostic contact lenses for patients with diabetes mellitus
(see second picture below).
- We are also developing the
material to sense glucose at higher concentrations, such as
found in blood. Successful sensor development has direct application
in monitoring glucose levels of inpatients in acute care situations,
both those with diagnosed diabetes mellitus and those who
experience acute care induced hyperglycemia.
- This technology has been
licensed for development by Glucose Sensing Technologies,
LLC.
Mass media coverage:
-
Chemical &
Engineering News, Sep9, 2002, p.43.
-
-
Scientific
American, April 16, 2003.
-
back
to list of projects
back
to top
Cancer
Protein Marker Sensing Material
-
There is intense
interest in developing in vivo sensors for clinically important
analytes and for markers of disease. We report here progress
in a program which seeks to create a new sensor material which
will be implanted under the skin to sample interstitial fluid.
-
This material
will be used to detect protein markers of cancer.
-
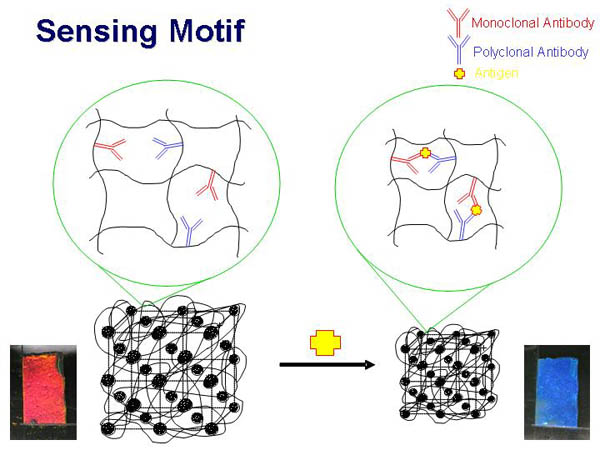
Our sensor
material consists of a crystalline colloidal array hydrogel
photonic crystal which will contain antibodies for these protein
markers. The signaling response utilizes the Bragg diffraction
of light by an embedded face centered cubic (fcc) array of
colloidal particles.
Back
to list of projects
back
to top
Organophosphate
Sensing Material
-
We have developed
an intelligent PCCA photonic crystal sensing material which
can sense organophosphate compounds at ultra-trace concentrations
in aqueous solutions.
-
A periodic
array of colloidal particles is embedded in an acrylamide
polymer hydrogel network, with a lattice spacing such that
it Bragg diffracts visible light.
-
The molecular
recognition agent for the sensor is the enzyme acetylcholinesterase,
which binds organophosphates irreversibly, creating an anionic
phosphonyl species.
-
The charged
species creates a Donnan potential between the interior and
exterior of the hydrogel, which causes an osmotic pressure
in the hydrogel network, increasing the gel volume . The volume
change increases the lattice spacing of the embedded CCA,
causing a red-shift in the wavelength of light diffracted.
-
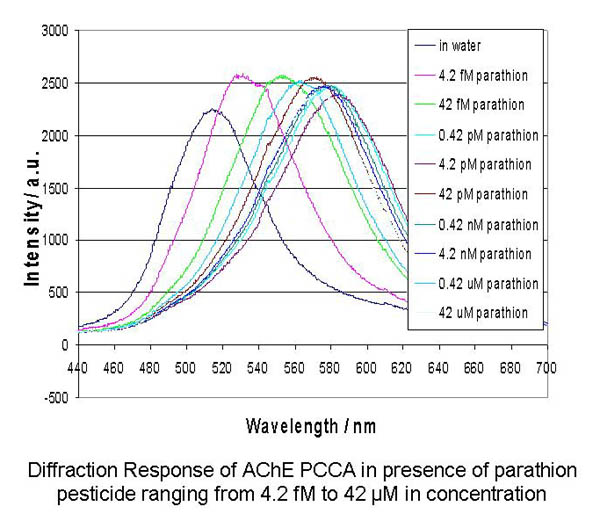
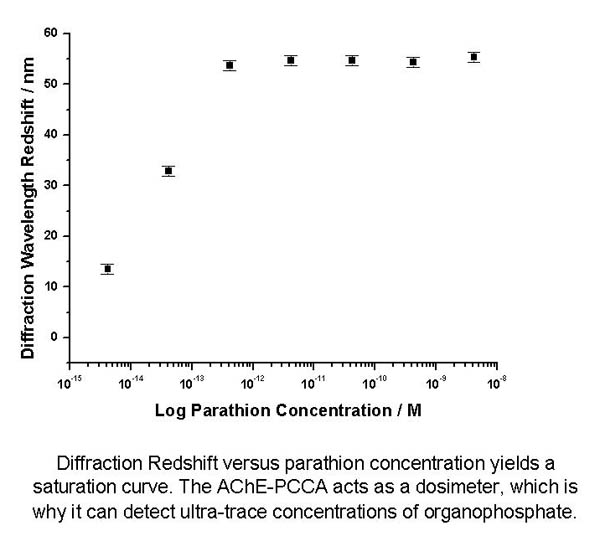
These AChE-PCCAs
act as dosimeters, as they irreversibly bind all parathion
in solution below stoichiometric ratios. The degree of redshift
in diffraction observed is proportional to the amount of bound
organophosphate. Parathion concentrations as low as 4.26 fM
are easily detected.
-
This sensor
can be used to determine ultra-trace concentrations of organophosphates
present in groundwater sources and soils. In addition, it
can be utilized with an air aspiration mechanism to detect
air-borne chemical warfare agents.
Back
to list of projects
back
to top
Point-of-Care
Ammonia Sensing Material
-
Ammonia arises
from deamination of amino acids. Hyperammonemia occurs in
it least four groups of inborn errors of metabolism: urea
cycle disorders, organic acidemias, fatty acid oxidation defects,
and liver malfunction. A point of care sensor capable of determining
ammonia concentration from 10 to 300 µM is valuable for diagnosis
and management of these patients.
-
We are developing
point-of-care test kit sensors for determining ammonia. The
sensors would also be applied to measure amino acid levels
through working with different deamination enzymes and subsequently
sensing ammonia.
-
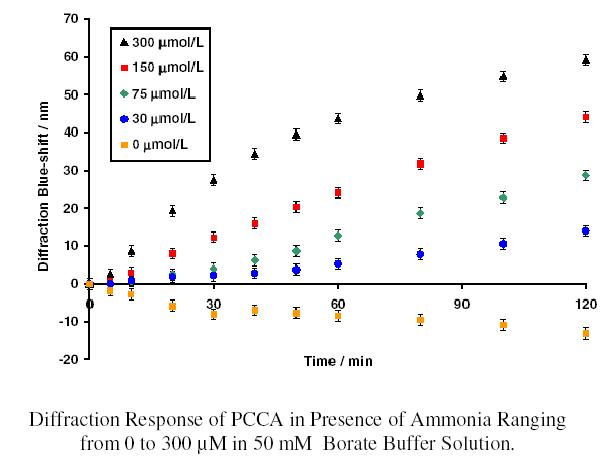
An ammonia
sensor, which utilizes the well-known Berthelot reaction,
has been developed in our group. Ammonia blue-shifts the PCCA
by forming crosslinks with two phenol groups tethered to the
hydrogel backbone.
-
We have currently
coupled glutamate dehydrogenase (GLDH) onto a pH sensitive
PCCA in the development of a second generation ammonia sensor.
The immobilized enzyme catalyzed reaction consumes H+, which
results in a red-shift of diffraction. We are working on improving
the sensor response to ammonia concentration under physiological
conditions.
Back
to list of projects
back
to top
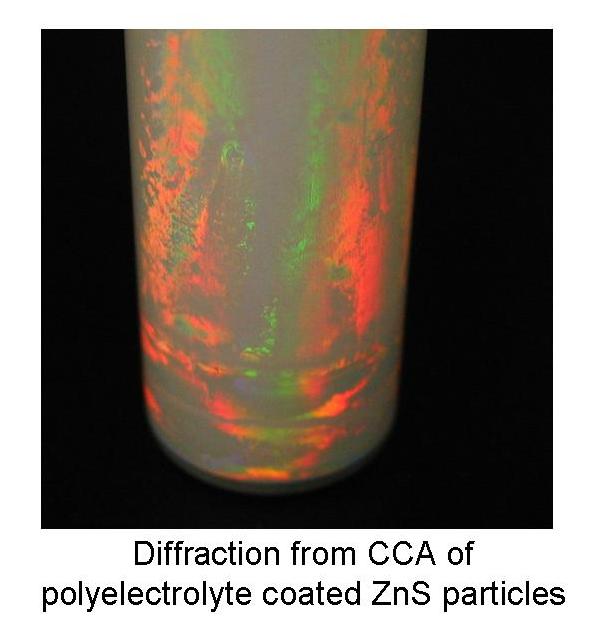
Zinc
Sulfide Particle Synthesis
- We have synthesized monodisperse
ZnS particles of various sizes between 100 NM and 500 NM and
coated them with polyelectrolyte molecules.
- These core-shell particles
are highly charged and self-assemble into a stable crystalline
colloidal array.
- This CCA demonstrates Bragg
diffraction peaks with unusually strong intensity and wide
band width, owing to the high index of refraction of ZnS (~2).
- It can be used in applications
such as coatings, filters and photonic crystal sensors and
devices.

Back
to list of projects
back
to top


![]() University of Pittsburgh
University of Pittsburgh![]()
![]()
![]()