 |
Assessment
of hs-CRP was applied to low-risk individuals in the Air Force/Texas Coronary
Atherosclerosis Prevention Study (AFCAPS/TexCAPS), a randomized, double-blind,
placebo-controlled trial of lovastatin in the primary prevention of acute coronary events.
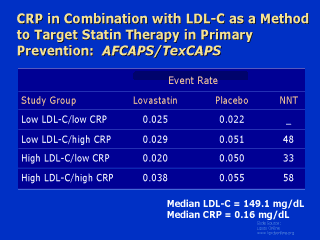
In this analysis, AFCAPS/TexCAPS patients were stratified into four groups based on their
LDL-C and their CRP levels. Those individuals with an LDL-C level above the study median
of 149 mg/dL had a clear benefit from lovastatin regardless of their CRP level; the number
needed to treat (NNT) to prevent 1 vascular event ranged from 33 to 58, a level at which
primary care intervention has previously been recommended. On the other hand, there were
marked differences between the two groups with below-median LDL-C levels. In those
patients with an LDL-C below 149 mg/dL but who had a high CRP level, the event rate with
placebo was just as high as in patients with LDL-C above 149 mg/dL, and most
interestingly, the efficacy of lovastatin was just as large. The NNT among individuals
with a below-median LDL-C level but an above-median CRP level was also well within
treatment guidelines. In marked contrast, those individuals with a below-median LDL-C and
a below-median CRP level had very low event rates, and there was no demonstrable efficacy
of lovastatin therapy at all. Thus, these data suggest that the combination of CRP
screening with LDL-C screening might well provide an improved method for predicting
efficacy of statin therapy in primary prevention. Reference:
Ridker PM, Rifai N, Clearfield M, Downs JR, Weis SE, Miles JS, Gotto AM Jr, for the Air
Force/Texas Coronary Atherosclerosis Prevention Study Investigators. Measurement of
C-reactive protein for the targeting of statin therapy in the primary prevention of acute
coronary events. N Engl J Med 2001;344:1959-1965.
Web
site |
